
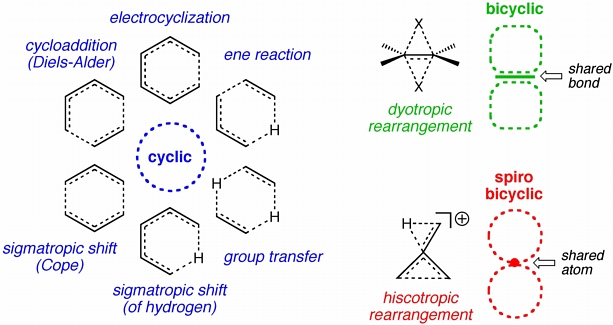
Pericyclic transition structures (examples are shown above in blue) involve the cyclic delocalization of electrons. Dyotropic transition structures (an example is shown above in green) can be thought of as the fusion of two sigmatropic transition structures such that they share a bond or connecting pi-system. Recently, we have happened upon another reaction type that combines two classic pericyclic reactions (an example is shown above in red). We call these "hiscotropic" reactions ("hisco" coming from a Latin word for "open" and "tropic" being a traditional word for "move" or "shift"), since transition structures for these rearrangement reactions can be thought of as the fusion of an electrocyclic (here, ring-opening) transition structure and a sigmatropic transition structure. We are particularly interested in reactions of this type where the two cycles of delocalized electrons share an atom, rather than a bond (i.e. are spiro-fused).
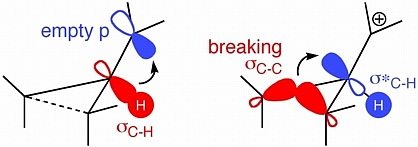
Our studies so far have focused on hiscotropic rearrangements of substituted cyclopropylcarbinyl cations. A generic representative of this reaction family is shown below in two views that highlight the key orbital interactions involved in controlling the structures and stabilities of spiro-fused hiscotropic transition structures.
Nouri, D. H.; Tantillo, D. J. J. Org. Chem. 2006, 71, 3686-3695: "Hiscotropic Rearrangements: Hybrids of Electrocyclic and Sigmatropic Reactions"
Tantillo, D. J. J. Phys. Org. Chem. 2008, 21, 561-570: "Recent Excursions to the Lands between Concerted and Stepwise: From Natural Products Biosynthesis to Reaction Design"
Hare, S. R.; Pemberton, R. P.; Tantillo, D. J. J. Am. Chem. Soc. 2017, 139, 7485-7493: "Navigating Past a Fork in the Road—Carbocation–π Interactions Can Manipulate Dynamic Behavior of Reactions Facing Post-Transition State Bifurcations"
Dustin H. Nouri and Dean J. Tantillo: "Hiscotropic Rearrangements." Poster presented by Dustin Nouri at the Bradford Borge Weekend, University of California, Davis, CA, February 24-25, 2006.
Dustin H. Nouri and Dean J. Tantillo: "Hiscotropic Rearrangements." Poster presented by Dustin Nouri at the SYLICCO.07 Symposium, Davis, CA, July 26, 2007.