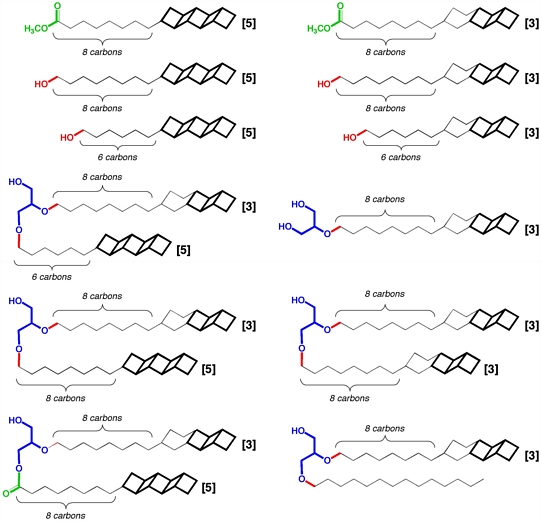
Ladderanes are hydrocarbons consisting of two or more fused cyclobutane rings. These molecules and their derivatives are of fundamental interest to physical organic chemists due to their strain and electronic properties. Much to the surprise of the chemical and biological communities, it was recently reported that a wide variety of ladderane lipids (above; ladderane substructures are bold, lipid headgroups are blue, red, and green) can be isolated from ammonium oxidizing bacteria living approximately 90 m below the surface of the Black Sea. That such highly strained polycyclic molecules are biosynthesized by Nature is both unprecedented and startling. The processes by which ladderane lipids are biosynthesized are not known and as of yet no enzymes that produce ladderanes have been characterized. We are using computational methods to assess the viability of several possible processes for biosynthesizing these concatenated cyclobutanes.
Nouri, D. H.; Tantillo, D. J. Curr. Org. Chem. 2006, 10, 2055-2074: "They Came from the Deep: Syntheses, Applications, and Biology of Ladderanes"
Nouri, D. H.; Tantillo, D. J. Tetrahedron 2008, 64, 5672-5679: "Sigmatropic Shifts and Cycloadditions on Neutral, Cationic, and Anionic Pentadienyl + Butadiene Potential Energy Surfaces"
Nouri, D. H.; Tantillo, D. J. Org. Biomol. Chem. 2012, 10, 5514-5517: "Attack of Radicals and Protons on Ladderane Lipids: Quantum Chemical Calculations and Biological Implications"
Hancock, E. N.; Kuker, E. L.; Tantillo, D. J.; Brown, M. K. Angew. Chem. Int. Ed. 2020, 132, 444-449: "Lessons in Strain and Stability: An Enantioselective Synthesis of (+)-[5]-Ladderanoic Acid"
Chen, S.-S.; Tantillo, D. J. ACS Omega 2020, 5, 26134-26140: "The Potential for Ladderane (Bio)synthesis from Oligo-cyclopropane Precursors"
Dustin H. Nouri and Dean J. Tantillo: "Ladderane Formation Mechanisms: The Uphill Battle." Poster presented by Dustin Nouri at the 232nd ACS National Meeting, San Francisco, CA, September 10-14, 2006; paper COMP 247.